Revelation Biosciences, Inc. Announces Financial Results For the Three and Twelve Months Ended December 31, 2025
SAN DIEGO – February 26, 2026 – Revelation Biosciences, Inc. (NASDAQ: REVB) (the “Company” or “Revelation”), a clinical-stage life sciences company focused on rebalancing inflammation to optimize health, today reported its financial results for the three and twelve months ended December 31, 2025.
Corporate Highlights
Announced Positive Results from PRIME Clinical Study in late-stage chronic kidney disease patients
Announced agreement with FDA on a single adaptive Phase 2/3 study for approval of Gemini for treatment of acute kidney injury
Received net proceeds of $6.7 million from warrant inducement in January 2026
Clinical data on the potential therapeutic benefit of Gemini for the treatment of acute kidney injury (AKI) and chronic kidney disease (CKD) will be presented at the International Conference on Advances in Critical Care Nephrology in San Diego (March 29 - April 1, 2026)
“2025 was a positive year for Revelation with significant advancement of the Gemini program," said James Rolke, Chief Executive Officer of Revelation. "We look forward to building on this momentum in 2026 to expeditiously bring Gemini to patients in need and adding to shareholder value."
Results of Operations
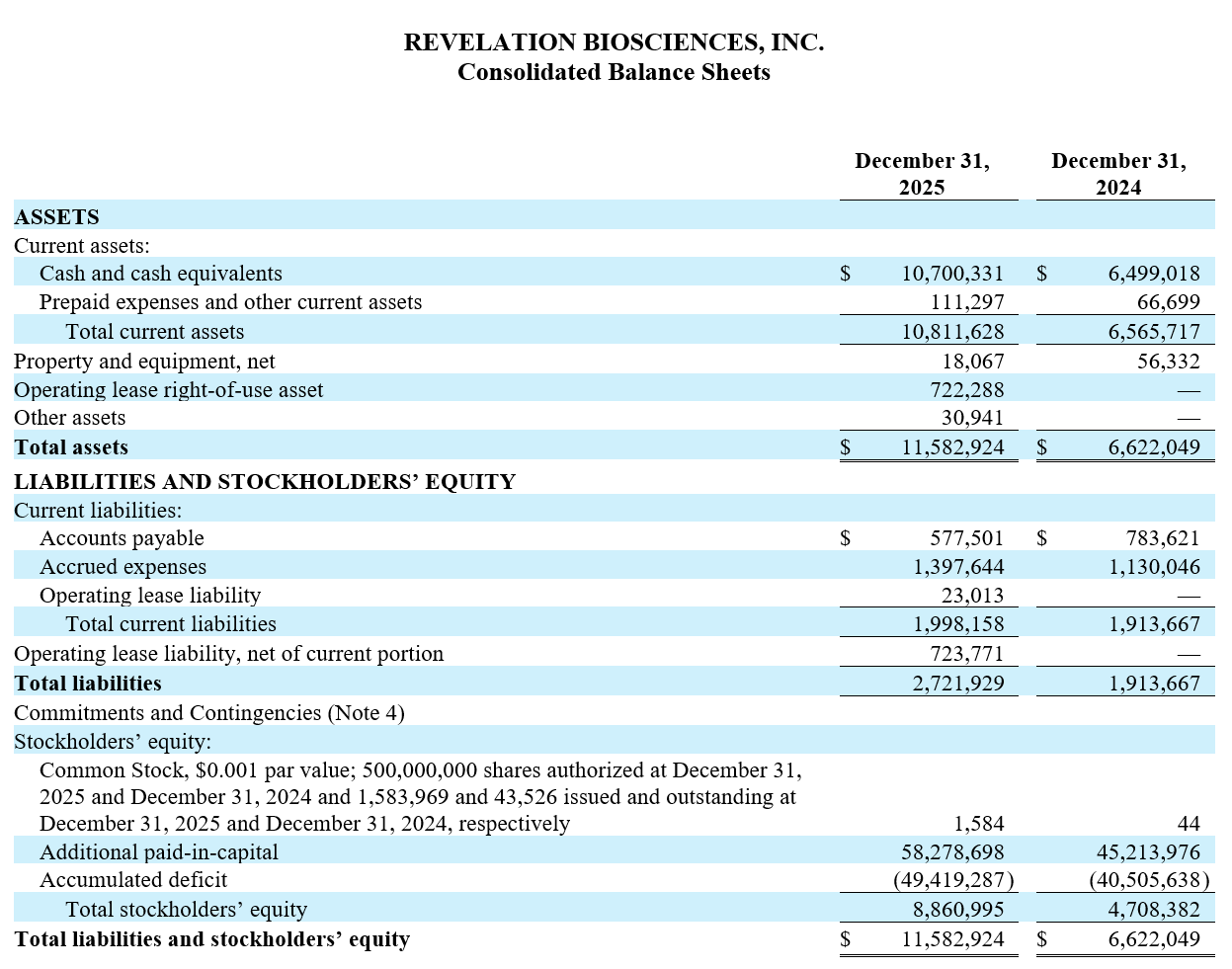
As of December 31, 2025, Revelation had $10.7 million in cash and cash equivalents, compared to $6.5 million as of December 31, 2024. The increase in cash and cash equivalents was primarily due to net cash proceeds from the May 2025 public offering and the September 2025 warrant inducement, offset by cash used for operating activities. Based on current operating plans and projections, Revelation believes its current cash and cash equivalents are sufficient to fund operations into the first quarter of 2027.
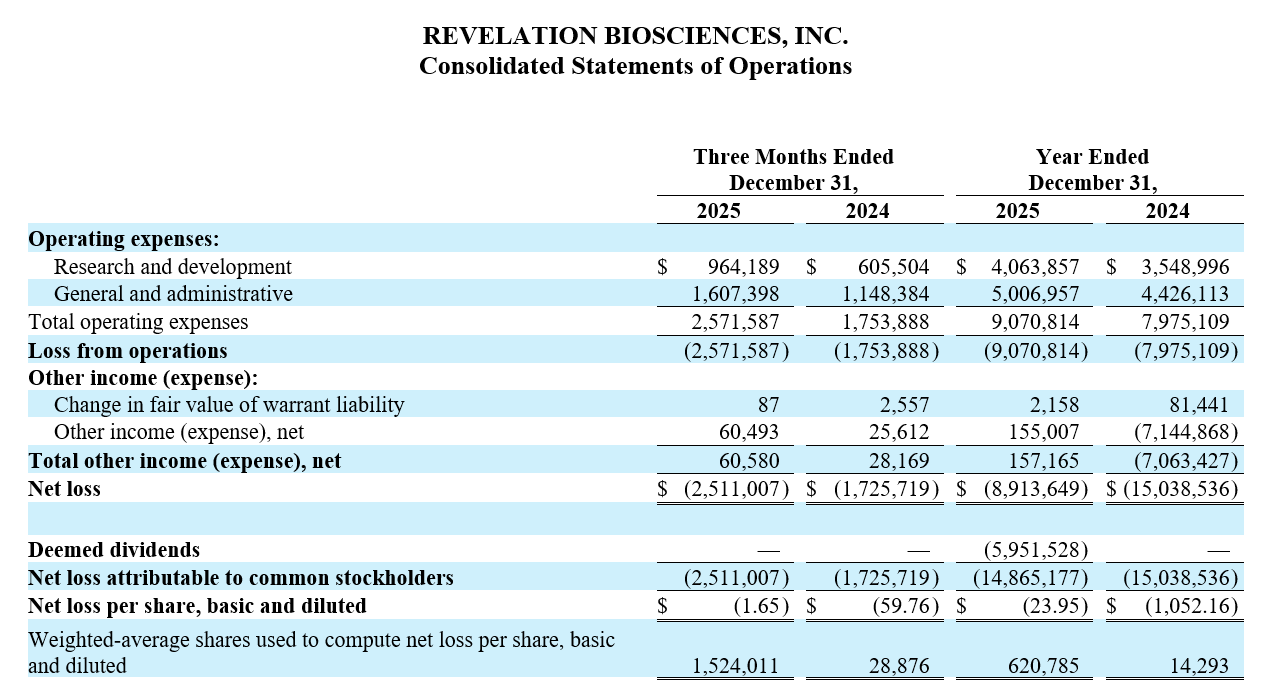
Net cash used for operating activities for the twelve months ended December 31, 2025 was $8.3 million compared to net cash used for operating activities of $18.3 million for the same period in 2024. Net loss for the three months ended December 31, 2025 was $2.5 million, or $(1.65) basic and diluted net loss per share, compared to a net loss of $1.7 million, or $(59.76) basic and diluted net loss per share for the same period in 2024. Net loss for the year ended December 31, 2025 was $8.9 million, or $(23.95) basic and diluted net loss per share compared to net loss of $15.0 million, or $(1,052.16) basic and diluted net loss per share for the year ended December 31, 2024.
About Gemini
Gemini is the Company’s proprietary formulation of phosphorylated hexaacyl disaccharide (PHAD®), a toll-like receptor 4 (TLR4) agonist. TLR4 stimulation with Gemini rebalances the innate immune response and has been demonstrated to have the potential to treat acute and chronic diseases associated with dysregulated inflammation. Gemini is currently being evaluated as a potential treatment for acute kidney injury (GEM-AKI); Gemini is also being developed as a treatment for chronic kidney disease (GEM-CKD), as a treatment to reduce hyperinflammation and infection associated with severe burn (GEM-PBI), and as a treatment to prevent post-surgical infection (GEM-PSI). The potential of Gemini to correct dysregulated inflammation has been demonstrated in multiple preclinical models of AKI, CKD, and infection, as well as in two phase 1 clinical studies. See additional detail here.
About Revelation Biosciences, Inc.
Revelation Biosciences, Inc. is a clinical stage life sciences company focused on rebalancing inflammation using its proprietary formulation, Gemini. Revelation has multiple ongoing programs to evaluate Gemini as a treatment for acute kidney injury, a treatment of chronic kidney disease, prevention of post-surgical infection, and a treatment to reduce hyperinflammation and infection associated with severe burn.
For more information, please visit www.RevBiosciences.com.
Forward-Looking Statements
This press release contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts. These forward-looking statements are generally identified by the words "anticipate", "believe", "expect", "estimate", "plan", "outlook", and "project" and other similar expressions. We caution investors that forward-looking statements are based on management’s expectations and are only predictions or statements of current expectations and involve known and unknown risks, uncertainties and other factors that may cause actual results to be materially different from those anticipated by the forward-looking statements. Revelation cautions readers not to place undue reliance on any such forward looking statements, which speak only as of the date they were made. The following factors, among others, could cause actual results to differ materially from those described in these forward-looking statements: the ability of Revelation to meet its financial and strategic goals, due to, among other things, competition; the ability of Revelation to grow and manage growth profitability and retain its key employees; the possibility that the Revelation may be adversely affected by other economic, business, and/or competitive factors; risks relating to the successful development of Revelation’s product candidates; the ability to successfully complete planned clinical studies of its product candidates; the risk that we may not fully enroll our clinical studies or enrollment will take longer than expected; risks relating to the occurrence of adverse safety events and/or unexpected concerns that may arise from data or analysis from our clinical studies; changes in applicable laws or regulations; expected initiation of the clinical studies, the timing of clinical data; the outcome of the clinical data, including whether the results of such study is positive or whether it can be replicated; the outcome of data collected, including whether the results of such data and/or correlation can be replicated; the timing, costs, conduct and outcome of our other clinical studies; the anticipated treatment of future clinical data by the FDA, the EMA or other regulatory authorities, including whether such data will be sufficient for approval; the success of future development activities for its product candidates; potential indications for which product candidates may be developed; the ability of Revelation to maintain the listing of its securities on NASDAQ; the expected duration over which Revelation’s balances will fund its operations; and other risks and uncertainties described herein, as well as those risks and uncertainties discussed from time to time in other reports and other public filings with the SEC by Revelation.
Company Contacts
Mike Porter
Investor Relations
Porter LaVay & Rose Inc.
Email: mike@plrinvest.com
Chester Zygmont, III
Chief Financial Officer
Revelation Biosciences Inc.
Email: czygmont@revbiosciences.com